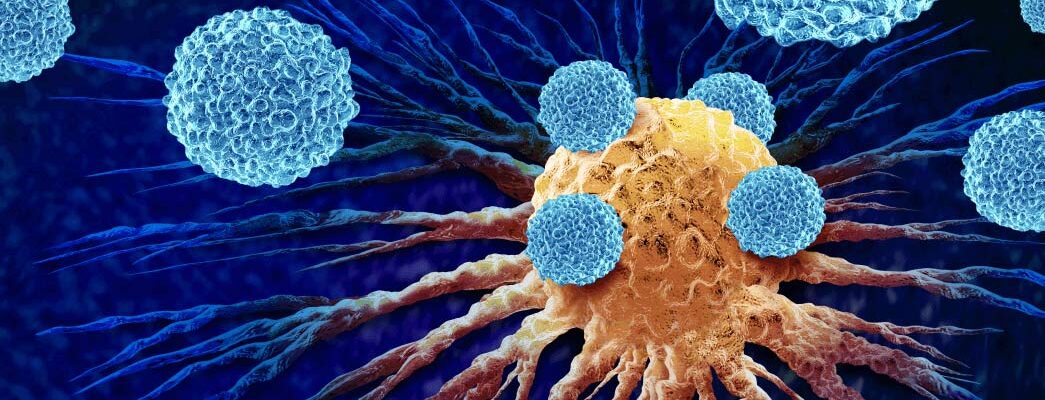
Mesothelioma is a rare and aggressive cancer affecting the thin lining around sensitive parts of the body including the lungs, heart, and abdomen. Because it is so aggressive, researchers are constantly looking for newer, more effective treatments.
T cell receptor fusion construct (TRuC) T cell therapy is one treatment currently being studied in clinical trials that is showing great promise. This treatment strengthens T cells’ ability to attack solid mesothelioma tumors and has demonstrated success in the early phases of research.
Learn more about what researchers say could be a favorable treatment to shrink mesothelioma tumors, even for patients who have had little success with other therapies.
What Is T Cell Receptor Fusion Constructs (TRuC) Therapy?
TRuC T cell therapy is an immunotherapy that uses modified T cells to target mesothelioma tumors.
T cells are critical cells that allow the immune system to target and kill dangerous infected cells. However, mesothelioma cancer cells can often sneak past the body’s natural defenses. By modifying T cells, immunotherapy stimulates the immune system fight off cancer cells.
Immunotherapy has been an exciting new development for multimodal mesothelioma treatment plans for several years, and TRuC T cell therapy, also called gavocabtagene autoleucel (gavo-cel), could join the list of treatment options because it can specifically target solid tumors that develop from mesothelioma and lung cancer.
How TRuC T Cell Therapy Targets Mesothelioma Tumors
Here’s a breakdown of how this new T cell therapy can target solid tumors:
- Doctors encode T cells: Through a blood sample, mesothelioma doctors remove T cells so they can be encoded with T cell receptor fusion construct (TRuC), which is a fusion protein made up of both the mesothelin antibody (MH1) and the T cell activator protein (CD3).
- Modified T cells detect mesothelin: With bolstered proteins, these T cells are reintroduced into the body to detect mesothelin(MSLN)-specific tumors and activate a strong attack against tumor cells to kill them.
- T cells continue to control cancer cells: For months after the initial injection of the T cells, they will keep immune system working against tumor cells, whether by preventing new cells from growing or fighting large tumors.
Results of TRuC T Cell Therapy Clinical Trial
A clinical trial led by researchers from the National Cancer Institute Center for Cancer Research examined how TRuC T cell therapy worked for 32 patients between May 2019 and May 2022. The trial participants included 23 mesothelioma patients, 8 ovarian cancer patients, and 1 patient with bile duct cancer.
Results from the trial showed:
- 30% of patients had a reduction in tumor size
- 20% were able to be evaluated for tumor response (after tumors had not responded to, on average, 5 other types of treatment)
- 13% had a long-lasting reduction in tumor size
Those with the most durable response saw results that continued up to a year after treatment.
One of the lead researchers, Dr. Raffitt Hassan, told the Center for Cancer Research, “This is one of the very few adoptive cell therapies in solid tumors that has shown objective tumor regressions, [proving] that TruC T cells work and we can get responses, even in heavily pretreated patients.”
One mesothelioma patient who had previously received 4 different types of treatment had a significantly positive improvement with TRuC T cell therapy. She experienced tumor response in just 5 days. Then, 64 days after treatment, her CT scan showed her tumor cells had been completely killed.
In phase II of clinical trials, Hassan and his team will fine-tune a second-generation TRuC that will still target mesothelin-specific tumors but also trigger additional immune responses that are sometimes shut down by mesothelioma.
Contact our Patient Advocates today so they can help find trials near you.
Risks & Side Effects of TRuC T Cell Therapy
Unfortunately, 78% of patients who participated in the phase I trial for TRuC therapy experienced cytokine release syndrome (CRS). This syndrome occurs when too many proteins responsible for immune responses are produced.
Common symptoms of CRS include:
- Body aches
- Diarrhea
- Fatigue
- Fever
- Nausea
- Vomiting
Also called cytokine-associated toxicity, CRS can be a potentially dangerous side effect if not caught quickly. However, it is typically a manageable condition. In this particular trial, CRS was the only negative side effect reported.
Find Clinical Trials for Mesothelioma Near You
Clinical trials can be an excellent way for mesothelioma patients to access some of the most promising emerging treatments. Still, it can be confusing trying to find the clinical trial that’s best for you.
Mesothelioma Hope’s team of nurses and Patient Advocates can connect you with top doctors and help you enroll in clinical trials that may help improve your life expectancy.
Contact us now at (866) 608-8933 to learn how we can help you along your mesothelioma treatment journey.